What a gout flare feels like clinically
It can start as a small, wrong-feeling twinge in a toe or ankle that seems out of proportion to anything you did that day. Then the joint can turn sharply tender—so tender that a bedsheet, a sock seam, or the edge of a shoe feels like pressure you can’t “tough out.” The skin may look red or purplish and feel hot, and the discomfort can peak quickly rather than building slowly.
One confusing part is how localized it can be: one joint acts like it’s on fire while the rest of you feels mostly normal, until the pain disrupts sleep or walking. Clinically, that pattern—sudden onset, intense point tenderness, warmth, swelling, and a preference for the big toe/foot/ankle—often fits gout, but it can still overlap with other causes of a hot, swollen joint, especially if fever, spreading redness, or injury is also in the picture.
Under the surface, the “shock” of the pain isn’t just swelling; it’s the joint’s immune response switching on fast. When urate crystals are present in the joint space, immune cells can treat them like a threat and release inflammatory signals that widen blood vessels and pull more cells and fluid into a tight capsule. That rapid crowding and chemical signaling is part of why it may feel sudden, inconsistent (fine at dinner, unbearable overnight), and hard to predict.
Uric acid buildup drives crystal formation
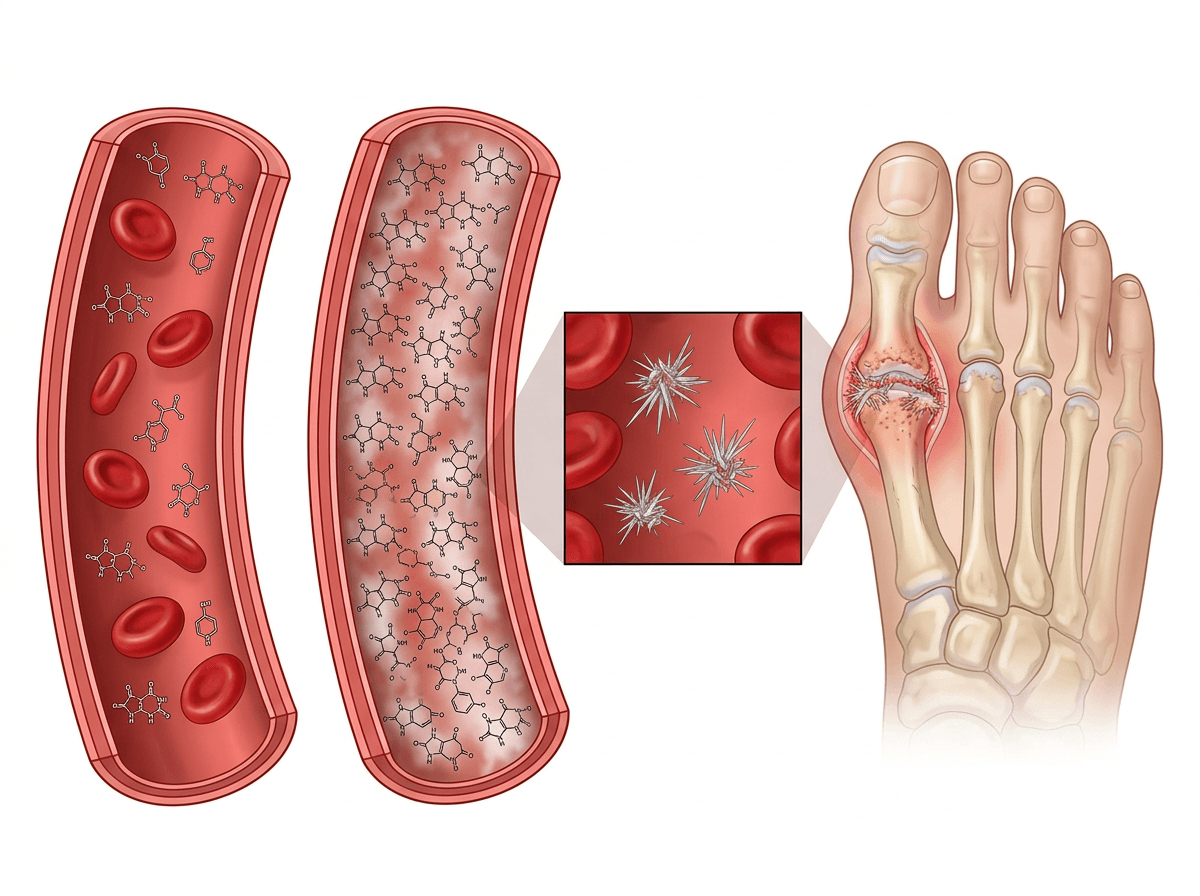
It’s often the “in-between” days that feel hardest to interpret—when the joint is quiet, but you can’t shake the sense that something is still going on in the background. That’s the part many flare medicines don’t touch: the level of uric acid circulating in the blood, which can rise or fall for reasons that aren’t obvious day to day.
Uric acid is what your body ends up with after breaking down purines, and most of it is supposed to leave through the kidneys. When production outpaces removal, the concentration can creep up—sometimes because kidney filtering is less efficient, sometimes because dehydration, certain diuretics, or other health conditions shift the balance. The frustrating inconsistency is that you can have high urate without pain, because inflammation doesn’t start until crystals form and the immune system notices them.
Once the level stays high enough, urate can come out of solution and form needle-like monosodium urate crystals, especially in cooler, lower-flow areas like toes and feet. That’s why a daily urate-lowering medicine is aimed at changing the “baseline chemistry” over weeks to months, even if it doesn’t feel like it’s doing anything during a sudden overnight flare.
Why crystals trigger sudden intense inflammation
Sometimes the oddest part is how quickly the joint seems to “flip” from mildly sore to untouchable, even if your uric acid issue has been building quietly for a long time. That speed can feel like it came out of nowhere, and it’s easy to assume something new must have happened overnight—when the bigger change is often that the immune system has suddenly decided those crystals are a problem.
When monosodium urate crystals sit in the joint space, they can be sensed by immune cells as a sharp, irritating signal rather than a neutral deposit. In response, those cells may release inflammatory messengers that open local blood vessels and make them leakier, which pulls more immune cells and fluid into a small, enclosed joint capsule. The space doesn’t stretch much, so pressure rises quickly, and the nerve endings in the area get amplified signals—part of why even light touch can feel disproportionately painful.
The stop-start nature is also part of the mechanism, not a personal failure to “figure out triggers.” In some cases, crystals can be present but relatively quiet until something shifts the local environment—like a small change in temperature, minor joint stress, or a change in urate levels that stirs crystals at the lining—enough for the immune response to escalate. That mismatch (crystals there for a while, inflammation suddenly loud) is why flare treatments focus on calming the immune reaction, not dissolving crystals in real time.
Fast-acting flare medicines and their targets
You can sometimes tell the “inflammation switch” is the real problem when the pain is still ramping up even though you haven’t done anything different—no new food, no new shoes, just a joint that won’t tolerate contact. In that moment, the medicines used for a flare are aimed less at urate itself and more at the chemical signals and immune-cell traffic that make the joint feel pressurized and hypersensitive.
NSAIDs (like naproxen or indomethacin) mostly work by reducing prostaglandins—messengers that amplify pain and blood-vessel changes—so the joint may feel less hot and less reactive, though stomach, blood pressure, and kidney limits can make the choice feel constrained. Colchicine targets immune-cell behavior (especially neutrophils), interrupting the “pile-on” response to crystals; it can be effective early, but the dose window can feel narrow because diarrhea or medication interactions show up fast for some people.
Corticosteroids (by mouth or injected into the joint) act more broadly, dialing down multiple inflammatory pathways at once, which is why they can help when NSAIDs or colchicine aren’t workable. The same wide net can matter more if you have diabetes, infection concerns, or frequent flares—situations where the short-term relief is real, but the context around it stays complicated.
Long-term urate-lowering medicines change the baseline
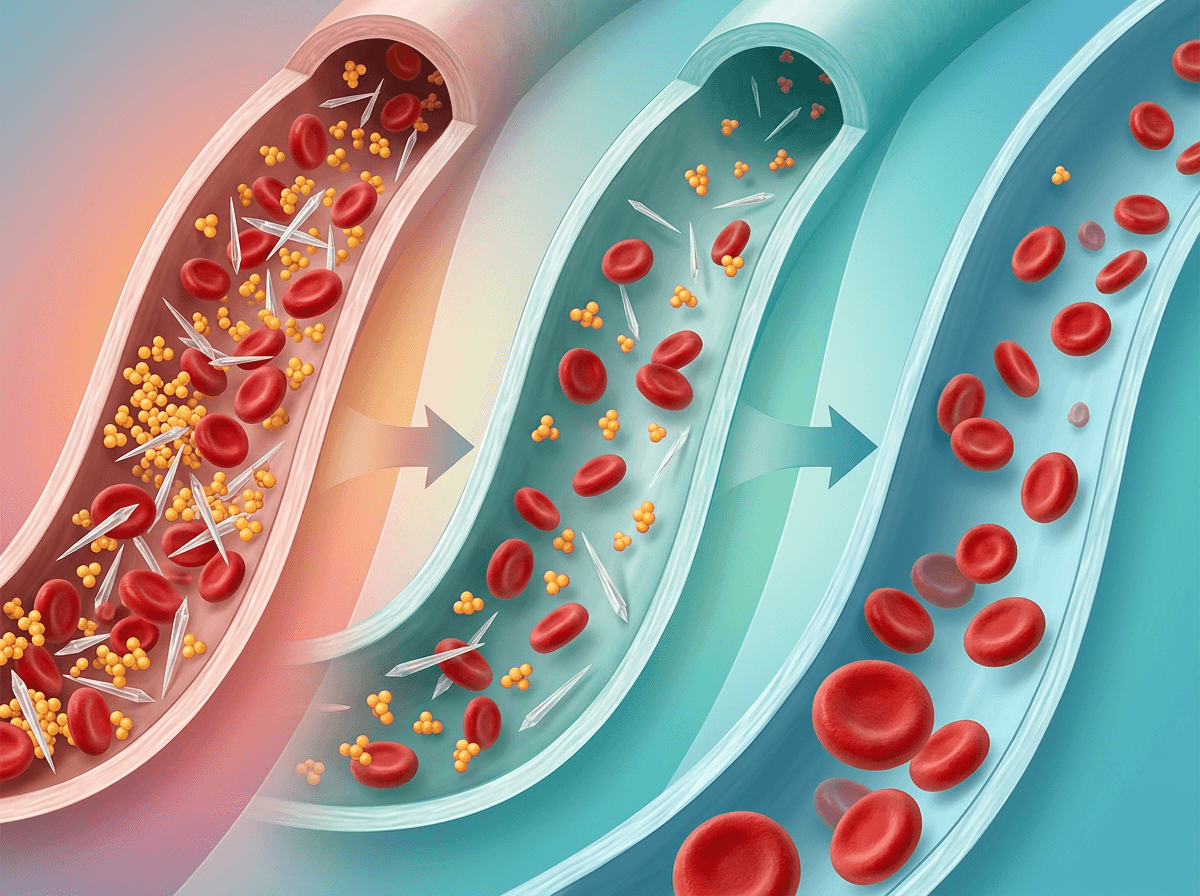
It’s subtle at first: the flare is over, the joint looks calmer, but your next lab result still feels like it’s “about a different body” than the one you’re walking around in. That disconnect is common because urate-lowering therapy isn’t designed to mute today’s swelling—it’s designed to change the concentration of uric acid over time so crystals are less likely to keep forming and hanging around.
Allopurinol (and febuxostat) work upstream by reducing uric acid production, so the bloodstream becomes a less crystal-friendly environment week by week, not hour by hour. Uricosuric medicines (like probenecid) work in the opposite direction, pushing the kidneys to excrete more urate, which can feel effortful to interpret if kidney function, hydration, or other prescriptions limit how well that pathway works. Either way, the goal is a slower baseline shift: fewer crystals getting added, and existing deposits gradually becoming less stable.
You can still have flares while that baseline is improving, because inflammation is an on/off reaction to crystals that may already be present. So “still flaring” doesn’t automatically mean the daily medicine failed—it can mean the chemistry is changing underneath while the immune system is still capable of flaring when crystals get disturbed.
Why starting prevention can briefly worsen flares
You might notice it most when you’ve done the “right” thing—started the daily urate-lowering pill—and then a flare shows up anyway, sometimes feeling sharper or oddly timed. That can be discouraging, because it reads like the prevention backfired, even though the chemistry is often shifting in a way that makes the joint temporarily more reactive.
As blood urate starts to fall, older crystal deposits in and around the joint lining can become less stable. In some cases they partially dissolve, shed, or break into smaller pieces, which means more crystal surface is exposed to immune cells. That can briefly increase the chance your immune system notices the crystals and triggers the same rapid inflammatory cascade you’ve felt before. The inconsistency is the hard part: labs may be improving while symptoms feel like they’re “resetting.”
This is why many clinicians pair the start (or dose increases) of urate-lowering therapy with short-term anti-inflammatory coverage, because the early phase is about getting through that churn while the baseline continues to move in the right direction.
Escalation options for refractory or complex gout
It’s the kind of pattern that makes you squint at your pill bottles: you’re taking a daily urate-lowering medicine, your numbers may be trending the right way, and yet the flares keep arriving on their own schedule. When that happens, the “next step” often isn’t a stronger version of the same flare medicine—it’s a closer look at whether urate is actually staying low enough over time, whether doses are being limited by kidney function or interactions, or whether the inflammation side is repeatedly getting re-triggered by lingering deposits.
In more refractory cases, clinicians may escalate by adjusting or switching urate-lowering therapy (for example, changing the xanthine oxidase inhibitor approach or adding a uricosuric pathway when appropriate), because lowering the baseline further can reduce the crystal load that keeps feeding the immune response. That can still feel inconsistent: the labs can look “better,” but the joint may remain vulnerable while deposits are being remodeled, especially if dosing changes are gradual for safety.
When standard options aren’t workable—because of kidney disease, medication conflicts, frequent severe flares, or tophi—care may broaden to specialist-directed strategies, including biologic therapies that target specific inflammatory signals, or in selected situations an intravenous enzyme that rapidly lowers urate. These are usually reserved for complex scenarios because the trade-offs (monitoring, infusion logistics, side-effect risk) are real, so it can help to frame the decision as balancing how disruptive the flares are against how intensive the treatment needs to be.











